|
REVISTA DE CHIMIE Cite as: Rev. Chim. https://doi.org/10.37358/Rev.Chim.1949 OSIM Nr. R102355 ISSN Online 2668-8212 ISSN Print: 1582-9049 ISSN-L: 1582-9049 |

LATEST ISSUE >>> |
| Volume 76, 2025 |
| Volume 75, 2024 |
| Volume 74, 2023 |
| Volume 73, 2022 |
| Volume 72, 2021 |
| Volume 71, 2020 |
| Volume 70, 2019 |
| Volume 69, 2018 |
| Volume 68, 2017 |
| Volume 67, 2016 |
| Volume 66, 2015 |
| Volume 65, 2014 |
| Volume 64, 2013 |
| Volume 63, 2012 |
| Volume 62, 2011 |
| Volume 61, 2010 |
| Volume 60, 2009 |
| Volume 59, 2008 |
| Volume 58, 2007 |
| <<<< back |
|
 During many of my professional and official trips in Europe and USA during 1990-1992, many scientists that I met (some of them Nobel Laureates) immediately after finding out that I am from Romania, their first question was Do you know Professor Balaban ? It was the easiest answer I ever responded to in my life. Yes, I do. But, I never told them how proud I was to be his undergraduate student at Polytechnic Institute from Bucharest in 1962, while he was a junior faculty in the Organic Chemistry Chair. Alexandru T. Balaban is the most productive chemist that Romania ever had, covering a large area of topics published 795 papers, 9 authored books, 79 books chapters, 8 edited books, and 27 patents. His outstanding intellectual skill was supported by a strong and permanently updated knowledge of chemical literature, and moreover he covered large and diverse areas of Chemistry. It is not my aim here to dive into too many technical details; those are nowadays easily available on public domains. I will rather emphasize, hopefully, in part the essence of Professor Balaban scientific achievement combined with a more sentimentalist approach on his superb chemistry and human traits. Some of my comments will be illustrated with excerpts from publications that bring some recollections from those passing years. During 60+ years of active involvement in chemistry research, Professor Balaban showed preference for both experimental and theoretical chemistry fields. However, the balance was modulated by objective justification (mostly regarding financial resources for chemicals, journals, books, instrumentation, etc.). A. Experimental chemistry 1. Reactions catalyzed by aluminum chloride. 2. Heterocyclic compounds: Pyrylium salts, oxazoles, boron compounds. 3. Free Radicals. B. Mathematical Chemistry: 1. Graph theory A1. Friedel-Crafts chemistry was the favorite topic for Professor Nenitzescu. Professor Balaban was admitted as graduate student (Professor Nenitzescu being his PhD mentor) in 1953, before finishing the undergraduate studies, and successfully defended his Thesis in 1959. The Thesis provided chemistry for 15 papers mostly published in peer review journals and two chapters in a Monograph. Two notable achievements have emerged from his Thesis. Contribution to understand the Scholl reaction (discovered in 1910), and lately used to synthesis of graphene nano ribbons. A Scholl intramolecular dehydrogenation is when 1-benzoylnaphthalene is converted by AlCl3 into benzanthrone. 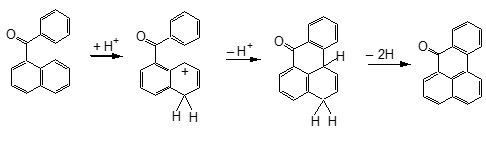 The expertise of Nenitzescu - Balaban team concerning AlCl3 reactions in Organic Chemistry, was quickly recognized internationally. Professor George Olah (Nobel Prize in chemistry in 1994) has invited Professors Nenitzescu and Balaban to write two chapters (Dehydrogenating Condensations of Aromatics (Scholl and Related Reactions) and Aliphatic Acylation), in the famous multivolume Friedel-Crafts and Related Reactions (1964). A2. The discovery of an efficient synthesis of pyrylium salts, presently known as Balaban-Nenitzescu-Praill reaction 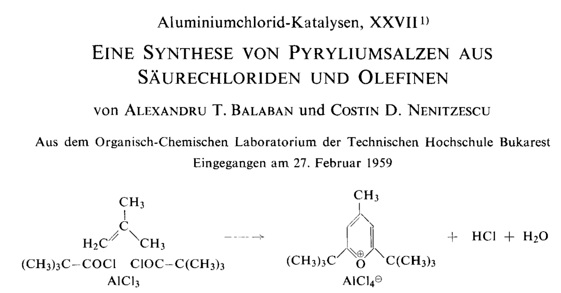 This is the most cited experimental chemistry paper of Professor Balaban (Web of Science accessed January 10, 2016): 125 hits.  A fascinating story, in the backstage of the PhD Thesis of Professor Balaban, that lead to the discovery of new synthesis of pyrylium salt is narrated in what follows. No references existed in the 70 years literature sources concerning the relative high distribution coefficient of pyrylium salt in water. The outstanding talent of the young chemist Balaban discovered this fact. While I started working in Professor Nenitescu's Organic Chemistry lab, I was fascinated to learn that Prof. Balaban had the idea to analyze the aqueous layer where pyrylium salts are soluble. Most of the biased organic chemist arrogantly neglected this. Traditionally, during an experimental work-up the attention is focused only upon the organic layer. This was a lesson I took home; for the rest of my professional life, I paid equally attention to the aqueous layer of the experimental work-up. Professor Balaban's discovery that alkenes can be diacylated to produce pyrylium salts continued after his PhD work to enlarge the scope of this chemistry. Many papers (see Rev. Roum. Chim. 2012, 57, 535-557) and review papers on this topics emerged from his pencil such as the first book on pyrylium salts: A. T. Balaban, A. Dinculescu, G. N. Dorofeenko, G. W. Fischer, A. V. Koblik, V. V. Mezheritskii and W. Schroth, Pyrylium Salts. Syntheses, Reactions and Physical Properties, Adv. Heterocyclic Chem., Suppl. Vol. 2, A. R. Katritzky (Ed.), Academic Press, New York, 1982 followed by - W. Schroth and A. T. Balaban, in "Methoden der Organischen Chemie (Houben-Weyl)", vol. E7b, Hetarene II (Teil 2), R. P. Kreher (Ed.), G. Thieme Verlag, Stuttgart, 1992; - T. S. Balaban and A. T. Balaban, "Pyrylium Salts" in "Science of Synthesis; Houben Weyl Methods of Molecular Transformations", Stuttgart, 2003; - A. T. Balaban and T. S. Balaban, in Science of Synthesis, Knowledge Updates 2013/3: Pyrylium Salts (Update 2013) m Thieme Verlag, 2013, Stutttgart, pp. 145-216 (see an excerpt below).  The interest for pyrylium chemistry, a domain in which Professor Balaban is so active and productive, has been extended in the recent years from research into the syllabus of the undergraduate experimental education. MIT Chemistry Department is one of the few places in the world where all chemistry majors undergraduate students are synthesizing a pyrylium salt, which in the next step is converted to N-t-butyl xylidine. In the last step of the synthetic sequence, the xylidine is incorporated into a molybdenum(III) complex. The three-coordinated molybdenum(III) complex is ultimately used to cleave the nitrogen molecule (see http://ocw.mit.edu/courses/chemistry/5-33-advanced-chemicalexperimentation-and-instrumentation-fall-2007/lecture-notes/n2lect.pdf) B. Mathematical Chemistry: chemical applications of graph theory During late 50' and early 60' Professor Balaban started to be interested in exploring if other atoms than can conserve aromaticity. In a systematic approach for all possible combinations of atoms capable to form monocyclic aromatic compounds, Professor Balaban concluded that boron-containing aromatic rings are worthy of a closer examination. Later, Professor Balaban became aware that his analysis was in fact the famous graph-theoretical called the necklace problem. Now a foot was set into starting a new domain: the graph theory. Frank Harary (the world's expert on graph theory) was instrumental in collaborating with Professor Balaban. Additionally, as a happy coincidence, the interest in Nenitzescu's group for <10-annulene> valence isomer and the invention by Prof. Balaban of reaction graphs for the reaction mechanism postulated by Professor Nenitzescu for a oxidation of hydrocarbons with Cr6+ provided the launching pad for a mature mathematical chemistry that exploded in the ensuing years. Now, we do not talk about Balaban reaction(s) but about Balaban index J, Balaban graphs (10-cage, 11-cage). In fact the most cited among his papers: 810 hits (Web of Science accessed on Jan. 9, 2016) is about Balaban index J: 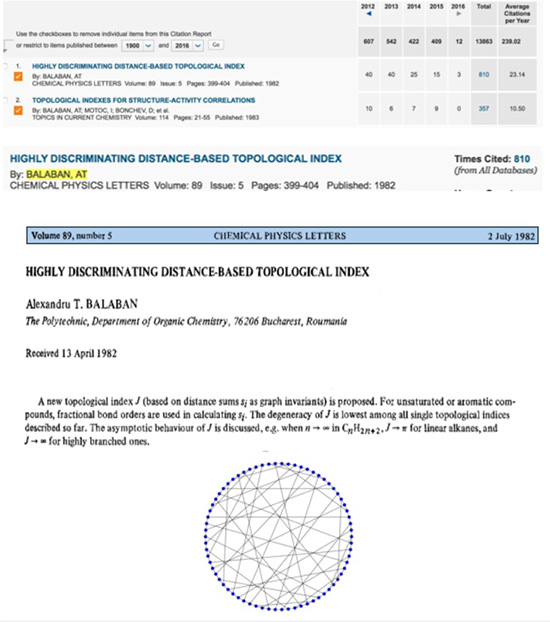 Balaban cage-10 is illustrated below: Many papers have resulted from an impressive international collaboration of Professor Balaban with chemists from USA (R. Hoffmann, NP 1985, D. J. Klein, P. v. R. Schleyer, C. Djerassi, J. A. Pople, F. Harary, A. R. Katritzky, W. Seitz), UK (A. R. Katritzky, P. F. G. Praill), Germany (W. Schroth, G. Fischer, V. Wray), France (C. Roussel, J. Metzger, C. Rulliere), Russia (V. I. Minkin, G. N. Dorofeenko) Croatia (M. Randiæ), Bulgaria (D. G. Bonchev, O. Mekenyan). Writing these lines, I cannot ignore the harsh conditions that any chemist in Romania, prior to 1989, was exposed to in each stage of a research producing science. Mostly during 80' there was lack of documentation (no journals, no books), no money for chemicals and major instrumentation, and extremely complicated and painful bureaucracy to publish. Only a few very strong and dedicated chemists have been able to survive. Definitely, our role model was Professor Balaban. Professor Balaban is a great teacher, all the time well prepared, clear in explanation and very eloquent in all comments. I still remember how hard I struggled to be in his Organic Chemistry Section. Is still vivid a personal experience after an un-announced Quiz with two topics, one about hybridization and a second water addition to the double bond. Because of my extensive readings of Pauling's original papers, I had the time to finish in one hour only the topics on hybridization. I had the pleasant surprise to see a 9+ grade (on scale 0-10) on my Quiz. Throughout my teaching carrier (50+) I also adopted the same attitude to highly grade a quality work even though the time limitations was precluding fully finishing the Exam. Vice-President of the Romanian Academy, decorated with National Order for Faithful Service by the President of Romania, proprietor of a very impressive scientific output, part of it created in an amazing broad international collaboration, awardee of the American Chemical Society (1994), tenured tracked as Professor at the age 70 at Texas A&M University at Galveston, Professor Balaban now is enjoying his 85th anniversary together with family (wife Nelly, children: Silviu, Irina, the grandson Alexandru; and Silvia, granddaughter) and many, many friends! Wishing you a Happy Birthday and at least as many more years as is the atomic number of Livermorium (Lv), named after the town of Livermore (Lawrence Livermore National Laboratory) where we presently live in California. Mircea D. Gheorghiu San Jose State University, CA 95192, USA Revista de Chimie, vol. 67 , no. 4, 2016 PDF Article |
| <<<< back |






